Run a trusted speak-up program in healthcare environments
Healthcare organisations operate in highly regulated, high risk environments. Whispli enables safe reporting across staff, patients and partners while maintaining confidentiality and regulatory compliance.




Trusted by industry
leaders worldwide
.webp)










.svg%20(1).avif)








.webp)
.webp)

.webp)





.svg%20(1).avif)








.webp)
.webp)

.webp)





.svg%20(1).avif)








.webp)
.webp)

.webp)





.svg%20(1).avif)








.webp)
.webp)

.webp)





.svg%20(1).avif)








.webp)
.webp)

.webp)


Why building a trusted speak-up program in healthcare is so difficult



Bring clarity, trust and accountability to healthcare reporting
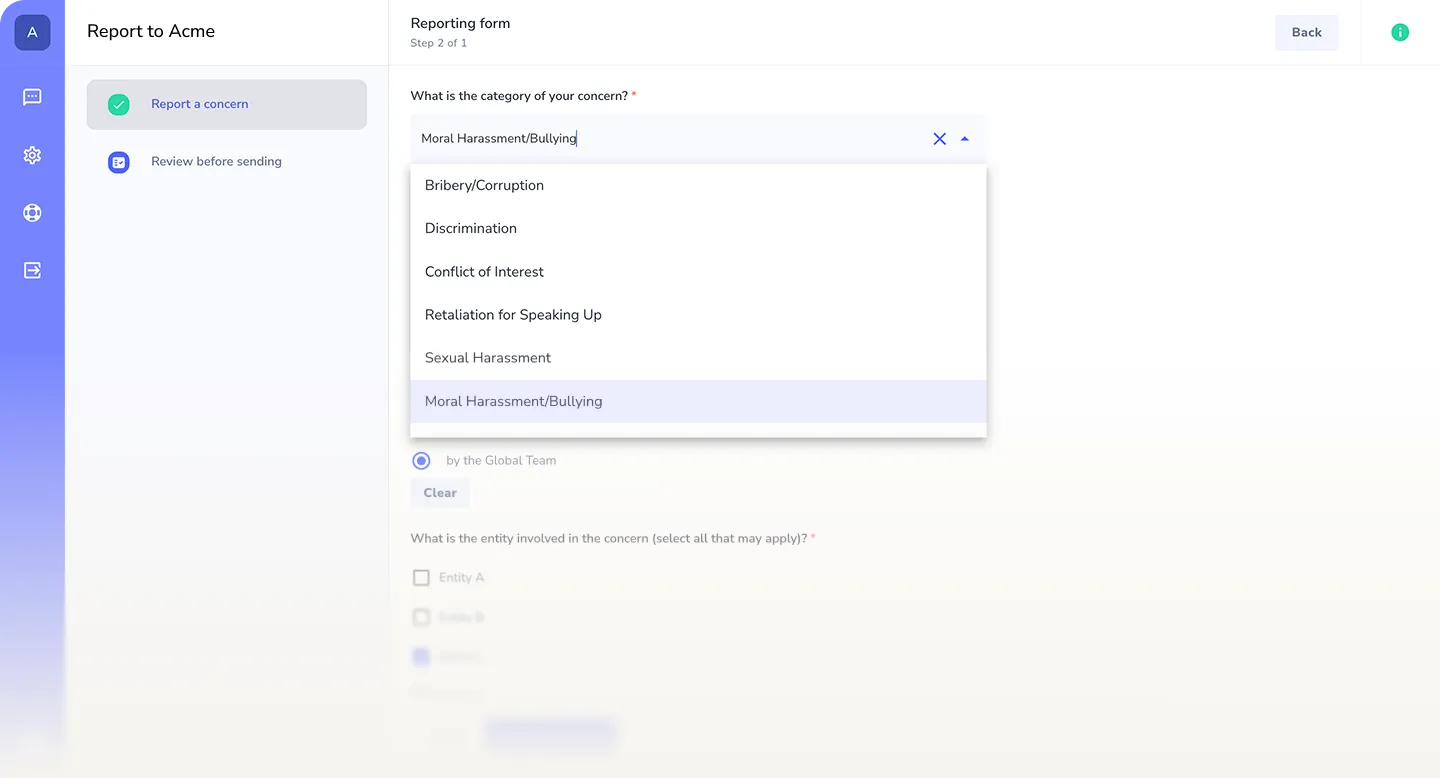


Built for healthcare compliance and duty of care

Support healthcare compliance obligations
Enable secure reporting aligned with healthcare regulations such as HIPAA, HITECH, GDPR for health data, EU whistleblower protection laws, and equivalent patient safety and data protection frameworks worldwide.

Protect sensitive health and personal data
Ensure the confidential handling of reports involving patient data, staff conduct, or clinical risks, in line with privacy, medical secrecy, and healthcare data protection laws.

Strengthen the duty of care and patient safety governance
Provide documented processes to identify, assess and address risks affecting patient safety, clinical practice, quality of care and ethical standards, including clinical governance and accreditation requirements.

Stay inspection- and audit-ready
Maintain clear records of reports, investigations and corrective actions to support audits, regulatory inspections, accreditation reviews and internal quality assessments with confidence.
Key capabilities for healthcare
organisations and regulated care environments

Multi-stakeholder reporting channels
Enable secure reporting aligned with healthcare regulations such as HIPAA, HITECH, GDPR for health data, EU whistleblower protection laws, and equivalent patient safety and data protection frameworks worldwide.
%202.svg)
Confidential handling of sensitive cases
Manage reports involving patient safety, clinical conduct, data privacy or workplace behaviour with strict access controls and case-level permissions.

Structured incident and investigation management
Capture reports in a consistent format, document investigations, corrective actions and outcomes across care units and facilities.

Support for patient safety and quality signals
Use reporting and trend analysis to detect recurring risks, unsafe practices or systemic issues affecting care delivery, including clinical governance, quality assurance and accreditation requirements.

Role-based collaboration across teams
Allow Compliance, HR, Quality, Risk and Medical teams to collaborate securely while preserving confidentiality and separation of duties.

Audit-ready traceability
Maintain complete records of reports, actions and decisions to support inspections, internal reviews and regulatory audits.
Outcomes healthcare organisations achieve with Whispli
Earlier identification of patient safety, clinical and compliance risks
Concerns related to care quality, data protection or workplace conduct are surfaced before they escalate into incidents.
Stronger regulatory alignment across healthcare environments
Reports are handled through structured, auditable processes aligned with healthcare regulations and internal governance.
Greater trust across staff, patients and partners
Confidential and consistent reporting reinforces confidence in speak-up processes across the organisation.
Empowering global organisations with higher engagement and stronger compliance outcomes
More than 300 companies, organisations and education institutions rely on Whispli to run their global speak up programs.
Whispli has been deployed in over 60 countries, demonstrating its flexibility and ease of configuration.
With no language barriers, Whispli empowers everyone to speak up confidently.

Modernise your global compliance strategy
Move from fragmented reporting tools to a single system of record designed for the realities of 2026.
Talk to our compliance experts and strengthen your global governance while uncovering risks before they escalate.
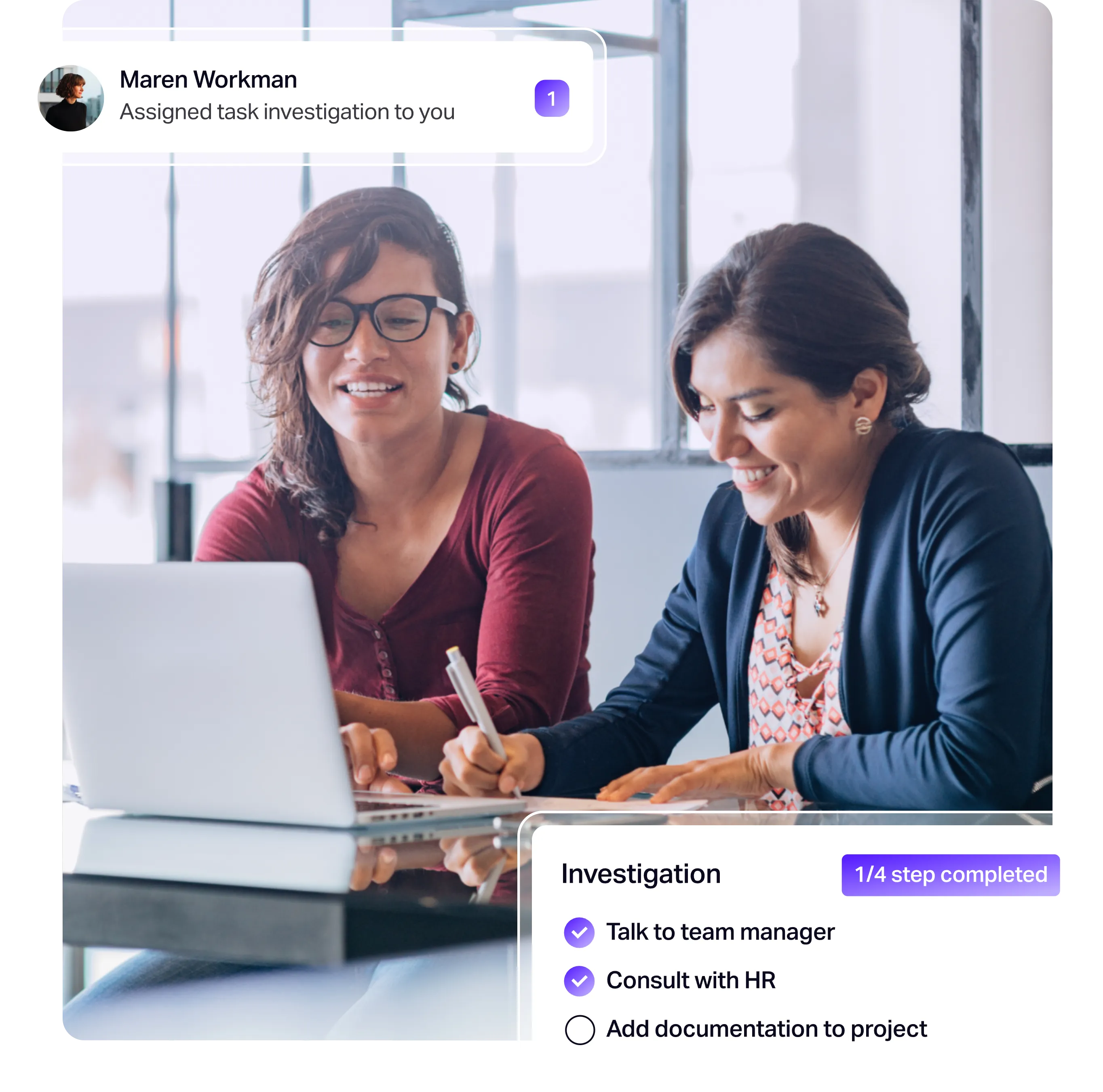
Explore more resources
Frequently asked questions
A structured speak-up program helps healthcare organisations identify ethics, safety and compliance risks early. It supports governance across clinical, operational, regulatory and administrative activities.
Reports may include patient safety concerns, data protection issues, clinical or research misconduct, harassment, conflicts of interest, fraud, or breaches of internal policies and codes of conduct.
Hierarchical structures, clinical pressure and fear of retaliation can discourage reporting. Anonymous reporting lowers barriers and encourages earlier disclosure of sensitive issues affecting patients, staff and compliance.
Yes. Whispli is used by a wide range of healthcare organisations, from hospitals and care providers to pharmaceutical and life sciences groups, adapting to different regulatory and operational requirements.














.webp)

.webp)
.webp)







%201.avif)
%201%20(2).avif)
%201%20(1).avif)
